Chemistry Game Board: The Chemisea
For the last four months, my class has been learning about the basics of chemistry. We have learned about the periodic table, ions, types of chemical reactions, etc. In October, we were given our first and final project of the first semester, where we had to create a game board for ages 10+ using what we had learned. The board required instructions, safety precautions, a blueprint, production of a gas, double and single replacement reaction, physical and chemical changes, and lighting an LED. So, we started building.
We started our project by selecting a theme, so we could create reactions based off of it. The theme we chose was 'Under the Sea.' I painted a board to look like the ocean, and it took me about four hours. I then drew stencils for the game path. I drew starfish, coral, crab, sand dollar, and a seahorse. The rest of the group then traced one animal nine times. Then, we cut out those animals and laminated then. We then, hot glued those squares into the path on the board. I then drew characters for players to choose as their game pieces. Then, Aaron and created instructions. The point of the game is to reach the treasure without triggering the chemical reactions along the game. These chemical reactions were made to look like fish. Landing on a space with one of the fish would trigger a reaction, and the players would have to move back one space.
The first reaction we constructed was a pufferfish, which used hydrochloric acid combined with calcium carbonate. We placed the acid in a flask and filled a balloon with calcium carbonate and stretched a balloon over the top. The balloon was then lifted so that the calcium carbonate would empty into the acid. This double replacement reaction resulted in carbon dioxide, which puffed up the balloon. We drew a pufferfish on it, so it looked like a pufferfish puffing up.
Our second reaction was a squid ink reaction, which was another double replacement reaction. It used silver nitrate and potassium chromate, which yields silver chromate and potassium nitrate. This reaction was also a precipitate. Silver chromate has a nice deep red color that looked like squid ink.
The third reaction was an angler fish. Aaron created an angler fish out of paper, and made the LED the light of the angular fish. The LED was then lit with a penny battery. To create a penny battery, you sand down one side of a penny, until the zinc under the copper is visible. Then stack pennies and acid soaked cardboard disks, until the stack can generate enough electricity to light an LED.
Our fourth reaction was a whale, which incorporated the elephant toothpaste reaction; a reaction that we had seen earlier in the year. Elephant toothpaste is a decomposition reaction. Hydrogen peroxide is combined with soap and slowly forms water and oxygen. A catalyst (we used potassium iodide) is used to speed up the process, and cause the whale to blow foam out of its blowhole.
Our final reaction was the underwater volcano. We used a Bunsen burner in our volcano, which is a combustion reaction. The methane gas from the burner combines with oxygen in the air, resulting in carbon dioxide and water vapor. We put cupric chloride into the fire, which made it burn green.
We started our project by selecting a theme, so we could create reactions based off of it. The theme we chose was 'Under the Sea.' I painted a board to look like the ocean, and it took me about four hours. I then drew stencils for the game path. I drew starfish, coral, crab, sand dollar, and a seahorse. The rest of the group then traced one animal nine times. Then, we cut out those animals and laminated then. We then, hot glued those squares into the path on the board. I then drew characters for players to choose as their game pieces. Then, Aaron and created instructions. The point of the game is to reach the treasure without triggering the chemical reactions along the game. These chemical reactions were made to look like fish. Landing on a space with one of the fish would trigger a reaction, and the players would have to move back one space.
The first reaction we constructed was a pufferfish, which used hydrochloric acid combined with calcium carbonate. We placed the acid in a flask and filled a balloon with calcium carbonate and stretched a balloon over the top. The balloon was then lifted so that the calcium carbonate would empty into the acid. This double replacement reaction resulted in carbon dioxide, which puffed up the balloon. We drew a pufferfish on it, so it looked like a pufferfish puffing up.
Our second reaction was a squid ink reaction, which was another double replacement reaction. It used silver nitrate and potassium chromate, which yields silver chromate and potassium nitrate. This reaction was also a precipitate. Silver chromate has a nice deep red color that looked like squid ink.
The third reaction was an angler fish. Aaron created an angler fish out of paper, and made the LED the light of the angular fish. The LED was then lit with a penny battery. To create a penny battery, you sand down one side of a penny, until the zinc under the copper is visible. Then stack pennies and acid soaked cardboard disks, until the stack can generate enough electricity to light an LED.
Our fourth reaction was a whale, which incorporated the elephant toothpaste reaction; a reaction that we had seen earlier in the year. Elephant toothpaste is a decomposition reaction. Hydrogen peroxide is combined with soap and slowly forms water and oxygen. A catalyst (we used potassium iodide) is used to speed up the process, and cause the whale to blow foam out of its blowhole.
Our final reaction was the underwater volcano. We used a Bunsen burner in our volcano, which is a combustion reaction. The methane gas from the burner combines with oxygen in the air, resulting in carbon dioxide and water vapor. We put cupric chloride into the fire, which made it burn green.
Reactions:
Pufferfish Reaction:
Type of Reaction: Double Replacement
Equation: CaCO3 (s)+ 2HCl (aq)→ CaCl2 (aq) + CO2 (g)+ H2O(l)
Squid Ink Reaction:
Type of Reaction: Double Replacement
Equation: AgNO3 (aq) + K2CrO4 (aq)→ AgCrO4 (s)+ KNO3 (aq)
Angler Fish Reaction:
How it Works: Each cell in a penny battery can produce up to 0.8 volt, and many can be stacked together to produce higher voltages. Since the battery is a wet cell, the effectiveness will be reduced when the electrolyte evaporates.
Elephant Toothpaste:
Type of Reaction: Decomposition
Equation: 2 H2O2(aq) → 2 H2O(l) + O2(g)
Volcano Reaction:
Type of Reaction: Combustion
Equation: 4CH4 (g)+ 2O2 (g) → CO2 (g) + 2H2O (g)
Each of These Reactions can be categorized as a physical and chemical change
Safety Precautions:
In the pufferfish reaction, hydrochloric acid is used. This chemical is coercive, and is an irritant to the eyes, skin, and mouth. When playing this game, please use gloves and goggles. If this acid gets into your eyes or on your hands, wash thoroughly with clean water.
In the elephant toothpaste reaction, hydrogen peroxide is used, and it is also coercive. Please follow the same safety precautions from above.
In the volcano reaction, a Bunsen burner is used, and can be dangerous. Please be aware of long hair, loose sleeves, and anything flammable around the open flame. If something does catch on fire, please use water or a fire extinguisher. If the flame is uncontrollable, please call the fire department and stop, drop, and roll.
In the elephant toothpaste reaction, hydrogen peroxide is used, and it is also coercive. Please follow the same safety precautions from above.
In the volcano reaction, a Bunsen burner is used, and can be dangerous. Please be aware of long hair, loose sleeves, and anything flammable around the open flame. If something does catch on fire, please use water or a fire extinguisher. If the flame is uncontrollable, please call the fire department and stop, drop, and roll.
Instructions:
The Chemisea is an underwater themed board game where two to four players race against each other to try and get to the sunken pirate treasure. Each player selects a character, either a mermaid, pirate, scuba diver, or lifeguard, and begins on the island. The eldest plays first. The first player selects a card from the deck and follows the instructions on the card. For example, the card could say, "Move forward three starfish," or "Move two spaces forward". As players move forward, they may land on a square covered in Xs. This means that they have triggered that square's corresponding reaction. The adults then mix the chemical for that reaction. After a player causes a reaction, he or she moves back one space, and the next player takes a turn. The first person to reach the treasure wins!
Follow the path down below.
Follow the path down below.
Set-Up:
Shuffle the cards and stack them together in a pile. Make sure all the cards are facing down, so players can't see what they will pick up off the top of the deck. Place the deck in the designated area on the board. Each player chooses one of the four characters. The eldest player goes first, with play continuing in a clockwise direction after that. Each player places his or her chosen piece at the beginning, chooses a card, and moves the number of spaces indicated on the card. If the piece lands on a field with X's, the player must go back on place. Play continues with the next player.
Materials:
-Wooden Board
-Acrylic Paint: Blue, green, yellow, red, brown
-Cardboard
-Newspaper
-Elmers Glue
-White Paper
-Painter's Tape
-Wooden Blocks for Support
-Bunson Burner and Tubing
-Methane Gas
-Lament
-Plastic Container
-String
-Paper Plate
-Acrylic Paint: Blue, green, yellow, red, brown
-Cardboard
-Newspaper
-Elmers Glue
-White Paper
-Painter's Tape
-Wooden Blocks for Support
-Bunson Burner and Tubing
-Methane Gas
-Lament
-Plastic Container
-String
-Paper Plate
Vocabulary:
Atom: An atom is the building block for everything. Everything on Earth is made of atoms. Atoms are formed from protons, neutrons, and electrons.
Element: An element is a piece of matter that is made up of only one type of atom. An example would be iron, which is made up of only iron atoms.
Compound: A compound is a molecule made up of multiple elements, so it has more than one type of atom. An example would be acetate, which is made of two carbon, three hydrogen, and two oxygen atoms.
Cation: A cation is an element or compound that has more protons than electrons. Metals are almost always cations.
Anion: An element or compound that has more electrons than it has protons. Anions have a negative charge. Non-metals are generally anions, with the notable exceptions of ammonium and hydronium.
Decomposition Reaction: The opposite of synthesis, where one reactant separates into two products
Combustion Reaction: A reaction where a hydrocarbon and oxygen react to form carbon dioxide, water, heat, and light
Single Replacement Reaction: A single replacement reaction is when a element swaps places with either the anion or cation portion of a compound. This takes the form of A+BC = B+AC, or A+BC = C+BA, depending on whether the element is a cation or an anion.
Double Replacement Reaction: A double replacement reaction is when two compounds in aqueous solution swap either cations or anions. This reaction only occurs if one of the compounds formed will be insoluble, according to the guidelines below.
Precipitate: The precipitate forms because the solid (AgCl) is insoluble in water
Catalyst: A catalyst is a substance that speeds up a chemicalreaction, but is not consumed by the reaction
Ions: Versions of an element that are not electrically neutral, meaning that they have a charge
Activity Series: The order of the reactivity of elements that allows us to predict whether a metal will displace the cation in a solution
Solubility Guidelines: Guidelines that allow us to predict whether two aqueous solutions will form a soluble solution or a precipitate
Chemical Change: A change in the chemical properties or composition of a material
Physical Change: A change in the shape, phase, or form of an object in which it retains its identity (composition, chemical formula, etc.)
Angular Fish: A scary fish with a light that acts as a lure to attract prey.
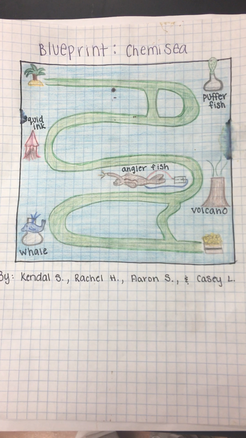
Blueprint:
Kendal drew a blueprint of the game to scale. The blueprint shows the path and the shortcuts.
Pufferfish:
Squid Ink:
Angler Fish:
Volcano:
Whale:
Conclusion:
This was one of our first real STEM projects, and it was pretty enjoyable. I had a great group, which consisted of Kendal, Aaron, and Casey. They were all so great to work with. It was easy figuring out a theme and splitting up the work. We started by figuring out the chemicals we wanted to use and how we could tie them into the theme. We then took inspiration from the instructions for Candy-Land for our own instructions. Working on the project was easy, but time management was were we got tripped up. We had to stay in for lunch a couple of days to finish. The night of our presentation went smoothly. The one thing I would improve on was my leadership; I should have taken initiative a couple of times to make the group work better. However, many people enjoyed our design, and I loved how it turned out. So, all of the stress was worth it in the end.